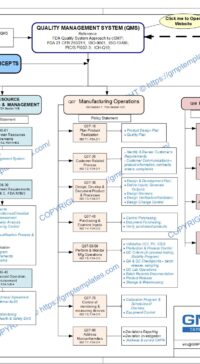
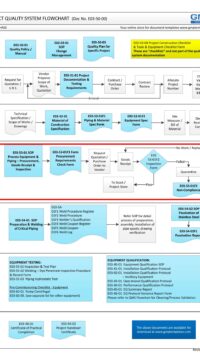
Description
Purpose of the Checklist:
The checklist consists of design criteria for a facility component that needs to be considered during the life-cycle phases of development, design, construction, testing, qualification and maintenance. This is to ensure the components in the facility are:
- “fit for intended purpose”,
- compliant to regulatory/statutory requirements,
- easy and safe to operate and maintain.
The checklist:
- consists of topics relevant to components in a facility eg process, utility, testing, security equipment; building components and fabrics; electrics and controls
- covers all project disciplines – Architectural, Ventilation/HVAC, Process Equipment, Process/Utilities, Electrical, Process Control and Automation
- includes design criteria which are typical industry standard
- the checklist ensures that you have covered all expects in the design, construction, testing and maintenance of components in a facility
- a missed item or design feature can lead to non-compliance; very expensive and cumbersome retrofit; plus project delays
- some items are extracted from statutory and regulatory requirements eg FDA CFR 211.42 Building/Facility
The entire checklist in our library includes:
| No. of items in each separate Checklist | Number of Items in Checklist |
|---|---|
| E20-52 Architectural | 120 |
| E20-53 HVAC | 70 |
| E20-34 Process Equipment | 155 |
| E20-55 Process & Utilities | 150 |
| E20-56 Electrical | 125 |
| E20-57 Process Control/Automation | 285 |
| TOTAL | 905 |
The “Sample Checklist” consists of only 60 out of the 900 items. It is available as a “Free Download” to allow you to sample some of the contents of the full versions.
The full versions are supplied in MS Excel for ease of entering data and sorting the items.