Description
What is the Resource Management Policy about?
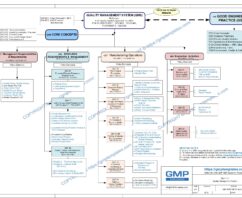
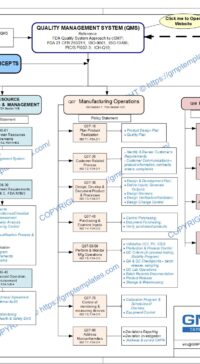
The four major sections in a Quality System Model are:
- Management Responsibilities
- Resource Management
- Manufacturing Operations
- Evaluation Activities
A Policy Statement defines the path for detail procedures and systems. The aim of this policy is to state the company’s policy to resource requirements and management to support the quality management system:
- ensuring that personnel performing work affecting quality is competent;
- the organization infrastructure (buildings, workplace, facilities, equipment and machinery, associated utilities, etc) is adequate;
- the work environment is capable to support the quality journey
Click to open and download an Adobe PDF format q00-00-01_qms_flowchart
Regulatory references:
- FDA Guidance for Industry – Quality Systems Approach to Pharmaceutical CGMP Regulations, September 2006, Section IV/B Resources
- ISO 9001:2000, Section 6 Resource Management Process
- PIC/S Guide to GMP for Medicinal Products, PE009-6, 5 April 2007, Chapter 2, 3 and 7
There are two parts to this document:
- Part 1 (2 pages) – Introduction to Template – describes the rationale of why this procedure or standard is required, cross reference relevant regulatory clauses.
- Part 2 (6 pages) – the document template.
Associated documents:
Q00-01 Quality Management System
Q06-01 Human Resources Requirements
Q06-30 Facility & Equipment