Description
What is a Site Master File for a Regulated Product, Facility and Business?
A site master file:
- is an overview of the company’s business and organization to produce and market quality products
- is a document prepared by the manufacturer containing specific and factual GMP information about the operational processes, production and/or control of pharmaceutical manufacturing operations carried out at the named site.
- A Site Master File for each manufacturing site listed in a product dossier, must be submitted to the relevant Regulatory Authorities.
- The contents of this document matches to the proposed guidelines and format by PIC/S. The document is fool-proof as it was written for a well established pharmaceutical facility – pick, choose and edit to suit your site and operations. The “Appendices” will prompt you for the relevant diagrams and information that need to be included.
There are two parts to this document:
- Part 1 – Introduction to Template – describes the rationale of why this procedure or standard is required, cross reference relevant regulatory clauses.
- Part 2 – the document template.

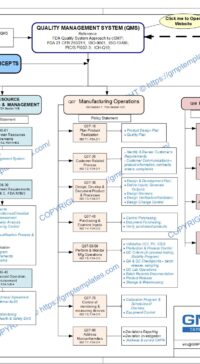
Management Responsibilities in Q05-0000 Quality Management System
Table of Contents:
1 GENERAL INFORMATION
2 QUALITY MANAGEMENT SYSTEM
3 RESOURCES (PERSONNEL)
4 PREMISES AND EQUIPMENT
5 DOCUMENTATION
6 PRODUCTION
7 QUALITY CONTROL
8 CONTRACT MANUFACTURE AND ANALYSIS
9 DISTRIBUTION, COMPLAINTS AND PRODUCT RECALL
10 SELF INSPECTION
11 APPENDICES