Description
What is this process and document about?
- This procedure provides a method to monitor and control engineering changes to process, equipment and utilities in a facility. This document is one of the procedures in Common Practices & Systems in GEP.
- The procedure and form define the sequence of initiating, authorizing, determining validation requirements and documenting the satisfactory completion of change. It is intended to ensure that all modifications of equipment with GMP impact are carried out in compliance with GMP, environmental, safety, and regulatory requirements.

E02-0000 GEP General-Practices
This document has 2 parts:
- Part 1 (4 pages)– describes the rationale why this procedure or standard is required, cross reference relevant regulatory clauses.
- Part 2 (9 pages)– the SOP document template + Form (3 pages).
Forms included:
Change Control Form (Engineering). This form steps through a checklist of:
- description, reasons and justifications of change
- Impact of change and affected departments
- Verification and evidence of work to implement change
- Concurrence and Close-out of change
Associated industry/regulatory guidance and requirements:
- ISPE Good Practice Guide: Good Engineering Practice, section 3.2
- ASTM E2500-7 Standard Guide for Specification, Design, and Verification of Pharmaceutical and Biopharmaceutical Manufacturing System and Equipment; Clause 8.4, 6.9.2, 7.1.4
- FDA (US) Q7A Good Manufacturing Practice Guidance for Active Pharmaceutical Ingredients, August 2001), Sections B3.
- PIC/S PE 009-8 2009 Guide GMP Medicinal Products – Part 1:5.23; Part 2:13
Included Sections:
Purpose, Scope, Training, References, Associated Documents, Definitions/Acronyms, Responsibilities, Procedures, Attachments, Form.
Who needs this document?
- Any department who uses equipment, instruments & utilities in R&D, manufacturing, testing, laboratories, logistics.
- Engineering/Projects department
- Quality Control
- Manufacturing
NOTE:
There are 3 Change Control templates in our website. The reasons for the three SOPs are due to their applications:
- E02-0201 CHANGE CONTROL (ENGINEERING) = most engineering changes occurs during the design stages prior to manufacturing and do not affect the “Critical Quality Attributes (CQA)” of the product. The SOP follows normal Good Engineering Practices (GEP) like revision control.
- Q04-0110 CHANGE CONTROL (this template) = follows all the steps expected from Regulatory Change Requirements – fully documented, executed and closed out by QA.
- Q04-0111 Document Change Control Procedure = this SOP relates to changes in GMP documents.
Related Documents or Topics:
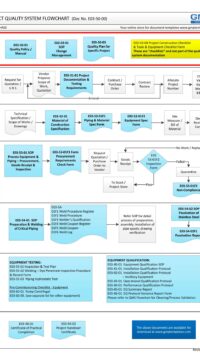
- E00-01-01 Policy Statement – Good Engineering Practice (also refer flowchart)
- E01-01-01 Risk Management
- E01-02-01 Cost Management
- E01-03-01 Organization and Control
- Q04-0110 Change Control (Quality)
This document belongs to the family – E02 Common Practices & Systems – Good Engineering Practice (GEP)