Description
What is a GMP Facility?
A production facility or clinical trial materials pilot plant for the manufacture of pharmaceutical products. It includes the manufacturing space, the storage warehouse for raw and finished product, and support laboratory areas.
What is GMP Project?
A project where the activities and deliverables are carried out in GMP environment and have the potential to affect the quality of the product manufactured in the facility.
What is this process and document about?
In order for a project to meet the expected outcomes, a quality management process helps to implement quality assurance and control measures. With the added regulatory requirements for a GMP Project, a Project Quality System is a must to deliver quality, compliance, cost and time benefits to a project.
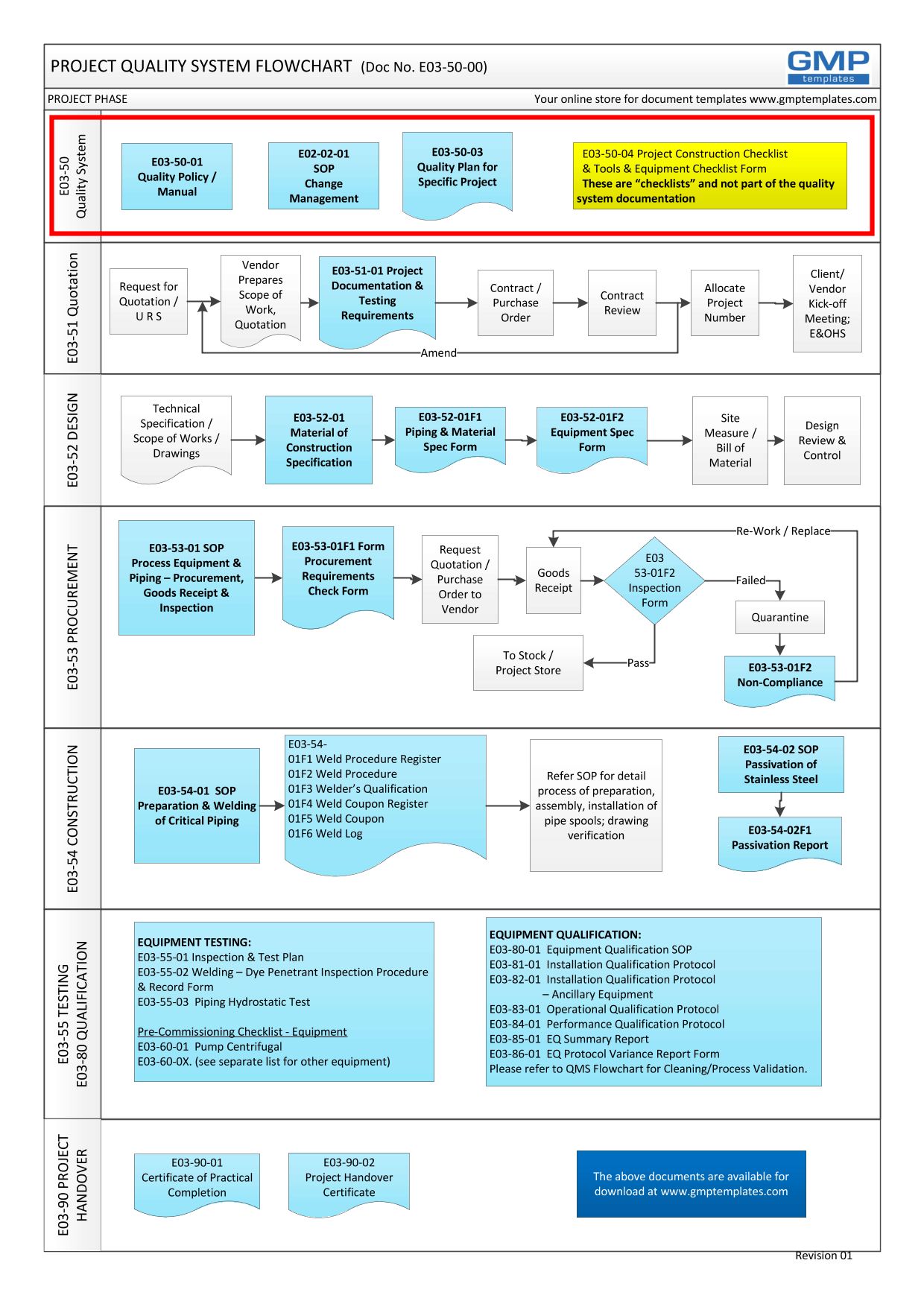
The Project Quality System covers the entire project GMP life-cycle:
- The core concepts of a quality system – eg Change Control have to be established at the fore front. This will set the expectation from all parties.
- Project Contract Stage – from the gathering of Design Inputs, request for quotation, contract review, etc. This is the relationship building between the Client and Contractor.
- Design Stage – from conceptual design to detail design and review; development of material of construction, equipment and piping specification.
- Procurement – acceptance criteria for goods inspection, addressing non-conformance
- Construction – SOP for stainless steel welding, installation, passivation
- Testing/Commissioning/Qualification – test plan and reports, IQ, OQ, PQ
- Project Handover process and reports
This document (Project Change Control) is one of the core concepts of the Project Quality System. The procedure and form will guide you through a register, change request, communication with stake-holders, impact/risk assessment, classification of change, safety & environment, testing requirements, close-out of change.
Click here to download an Adobe PDF version of Project Quality System Model
Attachments & Forms included:
Project Change Control Form
Associated Industry/regulatory guidance and requirements:
The following are only some of the references:
- ICH Q10 Pharmaceutical Quality System
- FDA Quality Systems Approach to Pharmaceutical CGMP Regulations
- Section – B/3. Facilities and Equipment – Under the CGMP regulations, the quality unit (QU) has the responsibility of reviewing and approving all initial design criteria and procedures pertaining to facilities and equipment and any subsequent changes (§ 211.22(c)).
- PIC/S PE 009-8 2009 Guide GMP Medicinal Products
- Part 1:5.23 ignificant amendments to the manufacturing process, including any change in equipment or materials, which may affect product quality and/or the reproducibility of the process should be validated.
- ASTM E2500-7 Standard Guide for Specification, Design, and Verification of Pharmaceutical and Biopharmaceutical Manufacturing System and Equipment
- 8.1 The specification, design, and verification process described should be supported by risk management, design review, and change management, as described in the following subsections.
- o 8.4.1 Change management processes should be established and be applied throughout the life-cycle
- ISPE Good Practice Guide: Good Engineering Practice (Core Concepts)
- Section 3.2.4 Planning and Monitoring, Sub-Practice 5: Change Management.Regulated companies should have systems for recognizing the need for change within project activities.
- the change management system should revise and amend the scope, schedule, and budget to reflect the selected outcome. The change and its potential impact should be communicated to the key stakeholders (e.g., users, quality department, project cost control, and senior management).
Keywords:
GMP Project Quality System Change Control
Who needs this document?
- Vendors – equipment fabricators and manufacturers;
- Contractors – Design & construct turnkey; mechanical piping contractors
- Projects, Validation, Engineering department
- Quality Control
- Manufacturing
Related Documents or Topics:
- E00-01-01 Good Engineering Practice (also refer flowchart E00-01-02)
- E01-01-01 Risk Management (FEMCA)
- E03-50-00 Project Quality System Flowchart
- E03-50-01 Project Quality System Policy
- E03-50-02 Project Change Control – this document
- E03-50-03 Project & Quality Plan (Concise)
- E03-50-04 Project Construction Checklist
- E03-51-01 Project Documentation & Testing Requirements
- E03-52-01 Material of Construction Specification
- E03-53-01 Process Equipment & Piping – Procurement, Goods Receipt & Inspection
- E03-54-01 Preparation & Welding of Critical Piping
- E03-54-02 Passivation of Stainless Steel & Record Form
EQUIPMENT TESTING & QUALIFICATION:
- E03-55-01 Inspection & Test Plan (Form)
- E03-55-02 Welding – Dye Penetrant Inspection Procedure & Record Form (E03-55-02F1)
- E03-55-03 Piping Hydrostatic Test Procedure & Form
- E03-55-04 SOP EQUIPMENT QUALIFICATION:
- -04F1 Installation Qualification Protocol – Ancillary Equipment
- -04F2 Installation Qualification Protocol
- -04F3 Operational Qualification Protocol
- -04F4 Performance Qualification Protocol
- -04F5 EQ Summary Report
- -04F6 EQ Protocol Variance Report Form
- E02-56-01 Project Handover – O&M Manual
- E03-56-01F1 Practical Completion Form
- E03-56-01F2 Project Handover Form