E03 PROJECT ENGINEERING
This sub-category of GEP relates to:
- E03-0000 Project Management practices and
- E03-5000 Project Quality System and procedures.
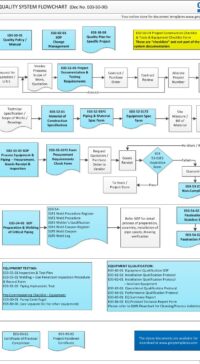
The templates are a must-have to ensure the smooth delivery of projects and the associated documentation. It is a well known issue within the Bio-Pharma community of the transition of GMP documentation between Engineering and Quality Unit.
The processes, templates and forms in this sub-category of GEP:
- In most cases, asking an Engineering Contractor to provide the “necessary GMP documents for the project scope” – will most often involve a lot of conflicts and misunderstanding of the “project scope”. The flowchart provides a roadmap to simplify the communication.
- provide a platform for a smoother project transition form Engineering to Quality Unit.
To view the E03-5000 Project Quality System flowchart in PDF – E03-5000_Project_Quality_Sys_Flowchart