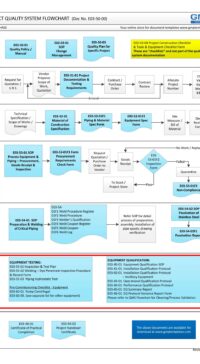
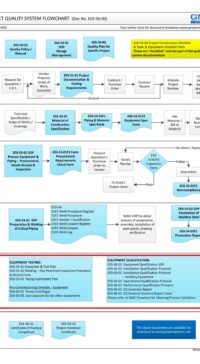
Description
Purpose of this SOP:
The purpose is to ensure all equipment and support systems decommissioning/removal are executed in a manner consistent with applicable codes, regulations, sound engineering practices and the GMP Life-Cycle model:
- GMP Facility & Equipment requires the associated GMP documentation to be closed out according to Site Quality Assurance.
- All safety aspects are addressed and approved by the site EH&S representative to ensure equipment is safe to be handled or re-used. This process ensures that equipment is decommissioned or removed in a manner that will allow the lowest possible risk to employees, operations or maintenance activities.
- Decommissioned equipment may be disposed of in the open market and re-used in a different environment and application. Equipment shall be fully cleaned and decontaminated to ensure residue is not present or present in quantities not detrimental to other processes. This is your legal obligation to ensure that the equipment/facility is safe to use by others.
The document template consists of:
- a 3-page Standard Operating Procedure
- a 5-page form with 25 prompts and checklist
Keywords:
Equipment Facility Decommissioning Asset Disposal Retirement
Who needs this template?
- Engineering, maintenance
- Contractors, service providers
- Production
- QA
- Validation
- QC
Associated documents and sections:
E02-23 Standards & Procedures – Construction
E04 Operations and Maintenance