Description
What is this procedure and document about?
Procurement is the process when materials and equipment are purchased, received, inspected and release for fabrication, assemble and installation. This procedure is one of the processes in a “Project Quality System”.
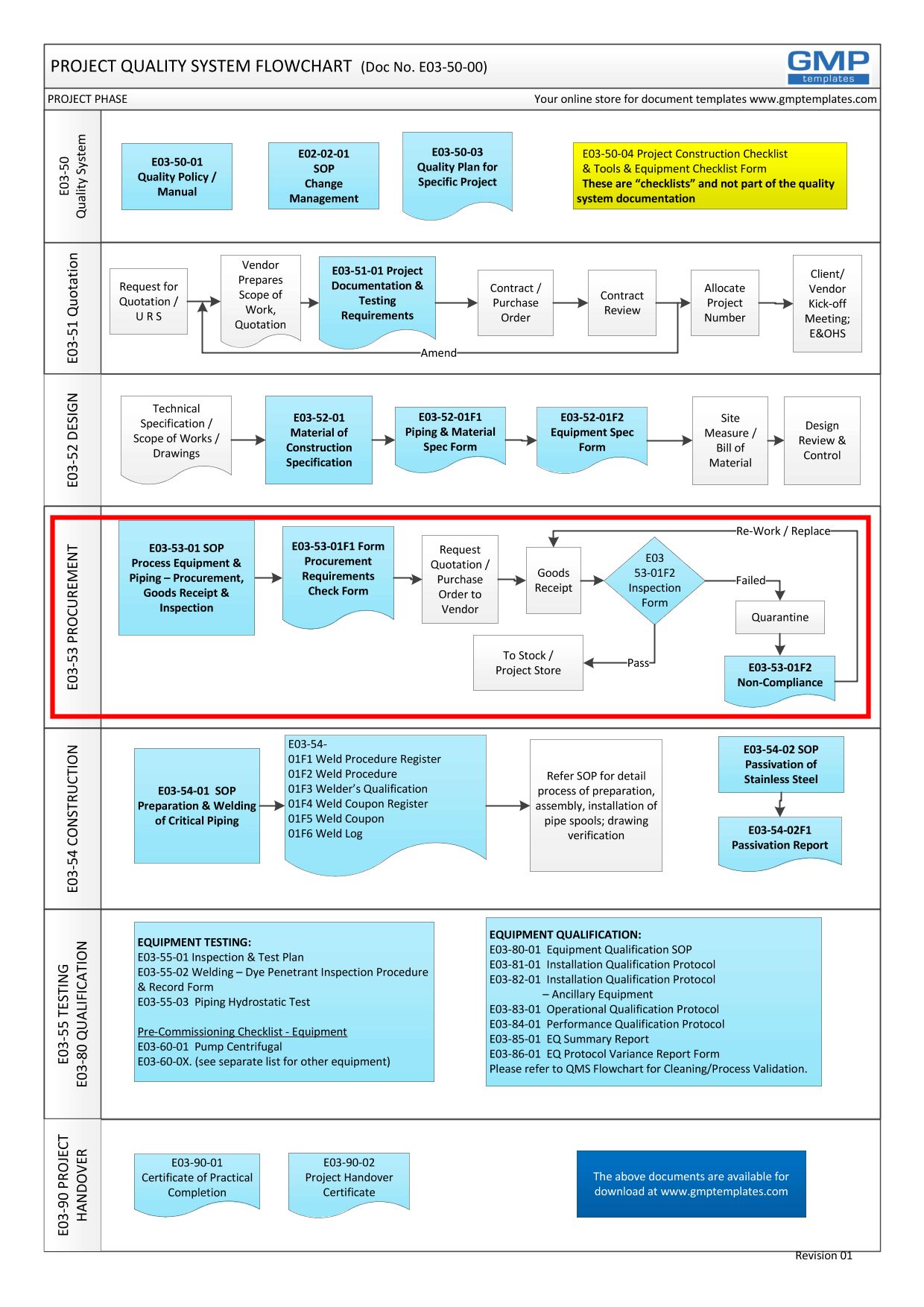
A Project may include all or in part the design, fabrication, installation, passivation, testing and qualification of process equipment and piping. A process map of a Project Quality System is shown above to guide you through each project phase from “Request for Quotation” to “Equipment Qualification”. To view the flowchart in PDF format or PRINT the flow chart – E03 Project Quality System Flowchart
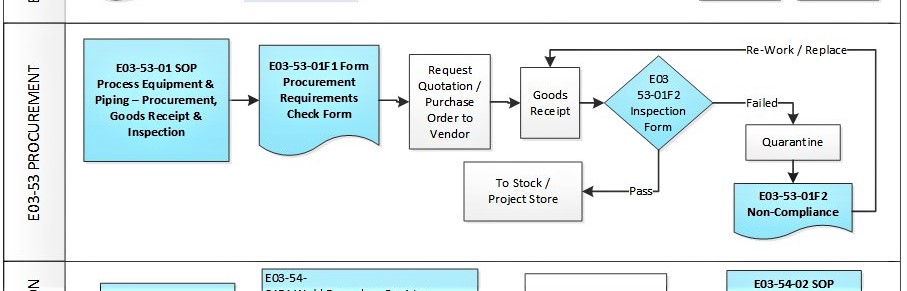
E03-50-00 Project Quality System Flowchart – Procurement
PURPOSE
The purpose of this document is to ensure technical specifications and acceptance criteria for a project are relayed to materials/equipment suppliers and sub-contractors:
- Employing the previously developed “DESIGN” processes and documents (eg technical specifications) to proceed to the next phase of a project – ie Procurement
- Procure materials, equipment, consumables and components process and ancillary systems (eg piping) for a project.
- Receive, store and inspection of incoming equipment and materials.
Included Sections:
- Part One – is an introduction to the document (2 pages)
- Part Two (6 pages) – the template has following sections – Purpose, Scope, References and Associated Documents, Responsibility, Procedures, Attachments (2 Forms)
Forms included:
- E03-53-01F1 Project Procurement Form
- E03-53-01F2 Project Inspection Form
An Adobe PDF version is available for Free Download E03-50-00_Project_Quality_Sys_Flowchart
Reference – Industry/regulatory guidance and requirements:
- ISPE Good Practice Guide: Good Engineering Practice
- ASTM E2500-7 Standard Guide for Specification, Design, and Verification of Pharmaceutical and Biopharmaceutical Manufacturing System and Equipment; Clause 6.6.3.3
Keywords:
Project Quality System, Process equipment and piping, material of construction, equipment qualification
Who needs this document?
- Vendors – equipment fabricators and manufacturers;
- mechanical piping contractors
- Any department who uses equipment & utilities in R&D, manufacturing, testing, laboratories
- Projects, Validation, Engineering department
- Quality Control
- Manufacturing
Related Documents (also refer Project Quality System flowchart):
- E03-50-00 Project Quality System Flowchart
- E03-50-01 Project Quality System Policy
- E03-50-02 Project Change Control – this document
- E03-50-03 Project & Quality Plan (Concise)
- E03-50-04 Project Construction Checklist
- E03-51-01 Project Documentation & Testing Requirements
- E03-52-01 Material of Construction Specification
- E03-53-01 Project Procurement & Inspection (this document)
- E03-54-01 Preparation & Welding of Critical Piping
- E03-54-02 Passivation of Stainless Steel & Record Form
EQUIPMENT TESTING & QUALIFICATION:
- E03-55-01 Inspection & Test Plan (Form)
- E03-55-02 Welding – Dye Penetrant Inspection Procedure & Record Form (E03-55-02F1)
- E03-55-03 Piping Hydrostatic Test Procedure & Form
- E03-55-04 SOP EQUIPMENT QUALIFICATION
- E02-56-01 Project Handover – O&M Manual