Description
What is this process and document about?
In order for a project to meet the expected outcomes, a quality management process helps to implement quality assurance and control measures.
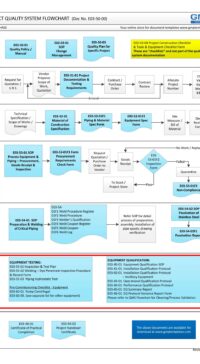
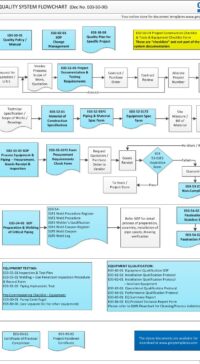
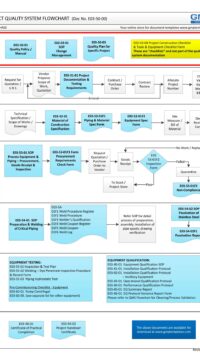
As part of the Project Quality System, this Project Documentation and Testing Requirements plays a major part in the entire project GMP life-cycle (see flowchart below) or click here to open in PDF format E03-50-00_Project_Quality_Sys_Flowchart
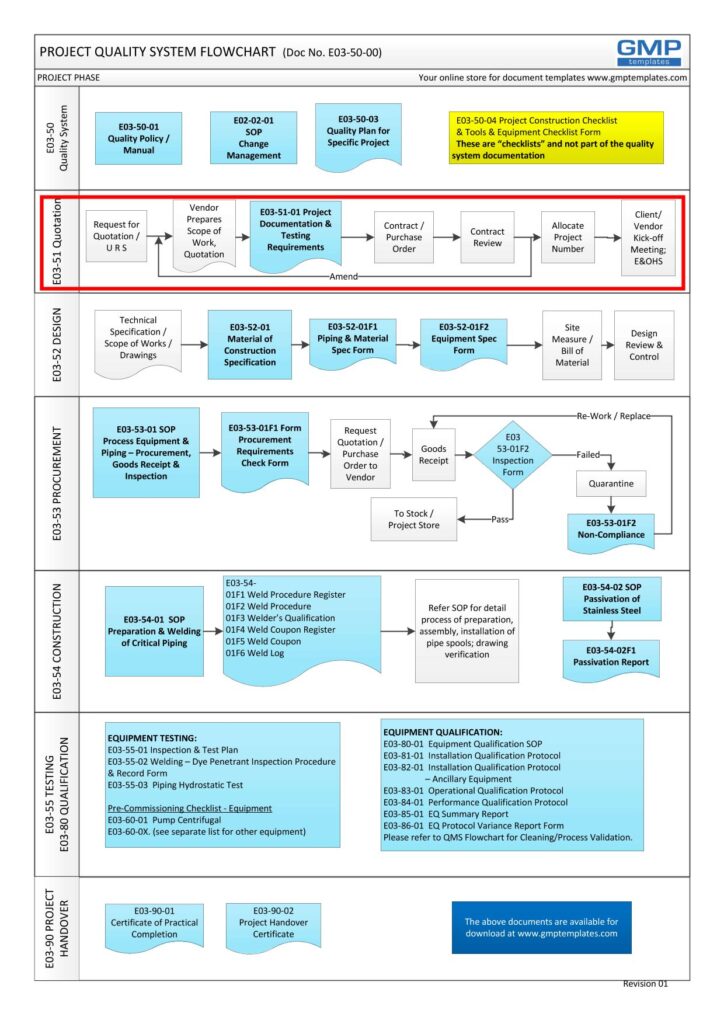
E03-5000_Project_Quality_Sys_Flowchart-2-Quotation
The documentation and testing requirements (including qualification) and the party responsible are stated upfront. This simple procedure/form combined (2 pages) will prompt the format and extend of the activities and documentation for qualification.
Keywords:
GMP Project Documentation and Testing Requirements
Who needs this document?
- Vendors – equipment fabricators and manufacturers;
- Contractors – Design & construct turnkey; mechanical piping contractors
- Projects, Validation, Engineering department
- Quality Assurance
- Manufacturing