Description
What is Equipment Qualification?
The design, construction and delivery of manufacturing systems and equipment into a facility requires a systematic approach in order to meet the user requirements and that of the relevant authorities like cGMP, local governing codes, law and regulations.
Equipment Qualification is the action of proving and documenting that the equipment and ancillary systems are properly installed, work correctly, and actually lead to the expected results. This is to ensure that all critical aspects or acceptance criteria for its intended use are met. Qualification is part of the validation process which is a regulatory compliance requirement.
The three components of Equipment Qualification are:
- Installation Qualification (IQ) – documented verification that the equipment or systems, as installed or modified, comply with the approved design, the manufacturer’s recommendations and/or user requirements. Source: PIC/S
- Operational Qualification (OQ) – documented verification that the equipment or systems, as installed or modified, perform as intended throughout the anticipated operating ranges. Source: PIC/S
- Performance Qualification (PQ) – documented verification that the equipment and ancillary systems, as connected together, can perform effectively and reproducibly based on the approved process method and specifications. Source: PIC/S
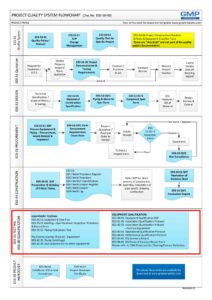
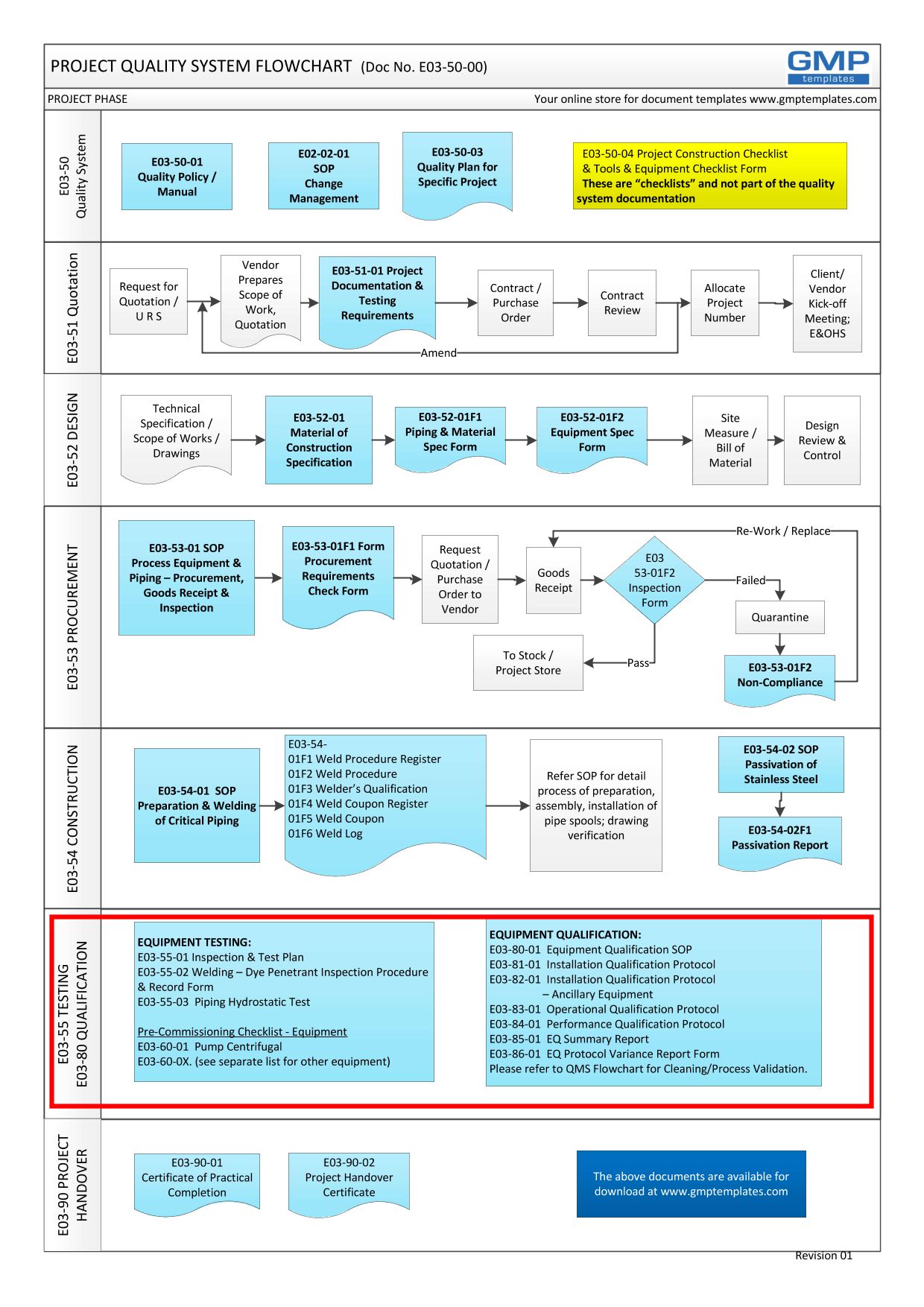
E03-50-00 Project Quality System Flowchart – Testing, commissioning and Qualification (Validation)
Click E03-50-00_Project_Quality_Sys_Flowchart to View-Download-Print the entire PDF flowchart.
What is this document about?
The purpose of this protocol template is to outline the tests required for Installation Qualification of a simple equipment
This protocol plays a role in two areas:
- Good Engineering Practice – it is part of the Testing process in a Project Quality System (see flowchart below)
- Quality Management System for a GMP facility – this protocol is a component of Facility & Equipment under Resource Requirements – View QMS Flowchart q00-00-01_qms_flowchart
This document was developed from a process improvement study of the validation process in a well established GMP facility. The qualification of a simple equipment should not required similar resource and effort to that of a complex equipment. This streamline 3 page protocol contains the sufficient tests to qualify an equipment as fit for intended use.
The protocol includes Forms:
- E03-80-01F1 Qualification Summary Report (4 pages) – this is a guide as to what should be included in a report
- E03-80-01F2 Protocol Variance Report Form (2 pages) – it guides you through a typical deviation investigation and CAPA process.
Associated Industry/regulatory guidance and requirements:
- ISPE Baseline Guide – Commissioning and Qualification
- FDA CFR Part 211 Subpart C (Building and Facilities) and Subpart D (Equipment)
- PIC/S Guide to GMP for Medicinal Products Part I (Clause 1.4/xi) and II (12.3 Qualification)
- ASTM E2500 Specification, Design, and Verification of Pharmaceutical and Biopharmaceutical Manufacturing System and Equipment
Keywords:
Equipment Qualification EQ Installation Qualification (IQ) Operational Qualification (OQ) Performance Qualification (PQ)
Who needs this document?
- Vendors – equipment fabricators and manufacturers; mechanical piping contractors
- Any department who uses equipment & utilities in R&D, manufacturing, testing, laboratories.
- Projects, Validation, Engineering department
- Quality Control
- Manufacturing
Related Documents:
E03-50-00 Project Quality System Flowchart
E03-50-01 Project Quality System Policy
E03-50-02 Project Change Control
E03-50-03 Project & Quality Plan (Concise)
E03-50-04 Project Construction Checklist
E03-51-01 Project Documentation & Testing Requirements
E03-52-01 Material of Construction Specification
E03-53-01 Project Procurement & Inspection Procedure
E03-54-01 Preparation & Welding of Critical Piping
E03-54-02 Passivation of Stainless Steel & Record Form
EQUIPMENT TESTING & QUALIFICATION:
E03-55-01 Inspection & Test Plan (Form)
E03-55-02 Welding – Dye Penetrant Inspection Procedure & Record Form
E03-55-03 Piping Hydrostatic Test Procedure & Form
E03-60-XX Pre-Commissioning Checklist – various equipment
E03-60-01 Pre-Commissioning Checklist – Pump (Centrifugal)
E03-80-01 Equipment Qualification SOP
E03-80-01F1 Qualification Summary Report
E03-80-01F2 Protocol Variance Report Form
E03-81-01 IQ Protocol Template
E03-82-01 IQ Protocol Template For Ancillary Equipment
E03-83-01 OQ Protocol Template
E03-84-01 PQ Protocol Template
E02-90-01 Project Handover – O&M Manual